By Brad Buecker, Senior Technical Publicist
Part 1 of this series began a discussion of makeup water treatment issues for the low-pressure boilers that exist at thousands of industrial plants around the globe. This part continues that discussion, with future posts focusing on good practices to minimize corrosion and scaling in boilers, and the feedwater and condensate return systems that supply them.
As noted in Part 1, sodium softening has served as the core of many makeup water treatment systems. However, the technology does not remove other ions from the water, including alkalinity, chlorides, sulfates, and silica, to name the most prominent. Several methods are available to improve makeup water purity beyond basic sodium softening. Some of the older, established technologies are:
- Split-Stream Dealkalization
- Hot Lime Softening
- Ion Exchange Demineralization
In large measure, these methods are from a bygone era, particularly the first two. The development and maturation of membrane technologies, and particularly reverse osmosis (RO) have altered the landscape. Single-pass, and especially two-pass RO, can produce makeup water with very low dissolved solids concentrations.
Figure 1. Cutaway view of an RO membrane
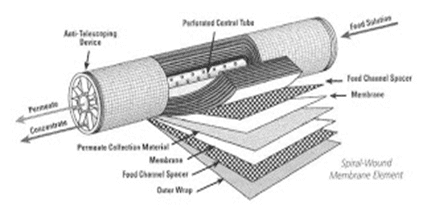
This Photo by Unknown Author is licensed under CC BY-NC
When pressure is applied to the makeup water feed to RO membranes (usually several series of membranes encased in individual pressure vessels), the membranes reject most of the dissolved solids (up to 99% or greater for modern membranes), to produce a significantly purified effluent (permeate) as makeup for the steam generator.
Keys to successful operation of RO units are pretreatment to remove suspended solids ahead of the RO membranes, and optimized chemical treatment to minimize scale formation on the membranes. Careful analysis of RO feedwater is critical for proper pretreatment equipment and chemical selection. Consultation with reputable RO suppliers and consultants is also necessary to select the proper design.
Use of higher purity makeup water as boiler feed can often reduce the complexity of boiler water treatment.
One major concern with systems whose makeup is treated primarily by sodium softeners alone is the accumulation of silica in the boiler. Silica chemistry, and its reactions with other elements, most notably magnesium and calcium, is rather complex. If insufficient hydroxide alkalinity is present in sodium-softened boiler water, and hardness breaks through from the softener, tenacious silicate scales may form in the steam generator. These insulating deposits reduce heat transfer and boiler efficiency, and may also lead to tube failures because of overheating. Protection from such scale formation is offered by high pH from hydroxide alkalinity, but this increases the potential for under-deposit caustic corrosion and potential tube failures from that mechanism. Minimization of silica transport to the boiler allows moderation regarding hydroxide alkalinity requirements.
Another concern, as has been hinted at before, is the presence of bicarbonate alkalinity (HCO3–) in boiler water from makeup systems with just sodium softeners. Some of this alkalinity reverts to carbon dioxide (CO2) at boiler temperatures, where the CO2 then flashes off with steam. The carbon dioxide can then re-dissolve in condensate return systems.
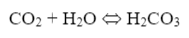
The resultant carbonic acid (H2CO3) lowers the pH of the condensate, which in turn can initiate general corrosion of carbon steel.
The upshot of this discussion is that more modern techniques are possible for makeup water preparation for low-pressure boilers. Like all other technologies, due diligence is necessary to determine the feasibility for utilizing these methods. Please consult all equipment manuals and guides, and, if you have any questions, reach out to our Technical Staff.
Click here to read part 3 of this blog series.
Please contact ChemTreat for assistance in designing a treatment program customized for your application. Like all other technologies, due diligence is necessary to determine the feasibility for utilizing methods. Always consult your equipment manuals and guides.

